
qScript XLT cDNA SuperMix
qScript Ultra Supermix is recommended as an alternative, offering improved performance and reduced reaction time.
Description
qScript XLT cDNA SuperMix is a next-generation tool for first-strand cDNA synthesis, providing a highly sensitive and easy-to-use solution for two-step RT-PCR and RT-qPCR. qScript XLT is an engineered M-MLV reverse transcriptase mutant with reduced RNase H activity and improved yield and stability at higher temperatures. Combined with a precise mixture of reaction components, this SuperMix enables superior results over a wide dynamic range of input RNA, with up to 8-fold higher sensitivity than our previous qScript cDNA SuperMix cDNA synthesis kits, which utilize an engineered RNase H(+) reverse transcriptase mutant.
Details
Single-tube, 5X concentrated reagent containing:
- Reaction buffer with optimized concentrations of molecular-grade MgCl2, dATP, dCTP, dGTP, dTTP
- Recombinant ribonuclease inhibitor protein (RIP)
- qScript XLT reverse transcriptase
- Titrated concentrations of random hexamer and oligo(dT) primer
- Proprietary enzyme stabilizers and performance-enhancing additives
Performance Data
Two-step RT-qPCR with high reproducibility, sensitivity, and broad dynamic range
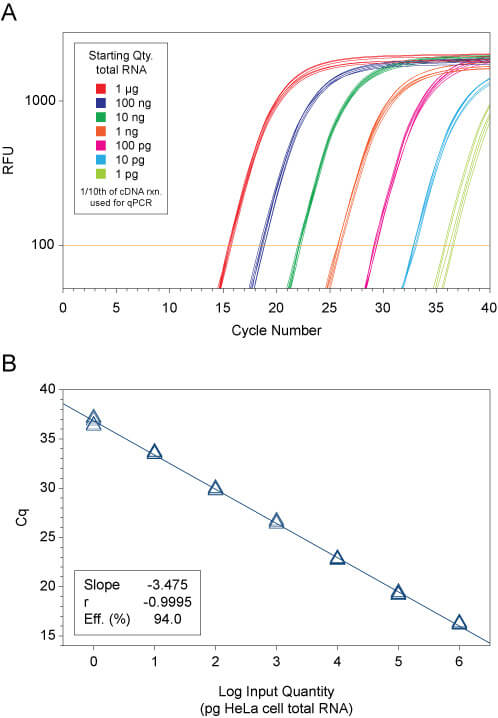
First-strand cDNA was synthesized using qScript® XLT cDNA SuperMix from varying amounts of HeLa cell total RNA (1 μg to 1 pg) . Six replicate cDNA reactions (20-μL final vol.) were performed for each input amount of total RNA template.
Following cDNA synthesis, 30 μL of 10 mM Tris (pH 8.0), 1 mM EDTA was added to each reaction and 5 μL of the diluted cDNA product (1/10th of each cDNA reaction) was used as template for qPCR s using PerfeCTa qPCR ToughMix with 0.5X Human B2M (FAM/MGB) TaqMan® Endogenous Control Assay (Life Technologies). qPCR was performed on a CFX96 Real-Time PCR Detection System (Bio-Rad). Following an initial activation step of 2 min. at 95°C, the 20-μL reactions were cycled 45X: 95°C, 5s; 60°C, 30s
Comparison of cDNA synthesis
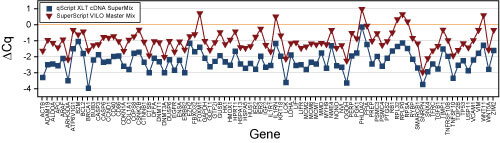
Comparison of cDNA synthesis from Brain tissue using qScript™ XLT cDNA SuperMix (Blue, Quanta BioSciences) and SuperScript® VILO (Red, Life Technologies).
Using Quanta’s original qScript cDNA SuperMix as a control, the dCq (change in cycle threshold) is shown for each of 96 genes.
The results indicate XLT beats VILO by at least a full cycle and the original SuperMix by more than 2 cycles on average.
Documents & Downloads
Customer Product Reviews
| 5 star | 100% | |
| 4 star | 0% | |
| 3 star | 0% | |
| 2 star | 0% | |
| 1 star | 0% |
 qScript XLT cDNA SuperMix
qScript XLT cDNA SuperMix

qScript XLT cDNA SuperMix was one of three cDNA synthesis kits I compared and I found it to be the best in terms of ease of use, cDNA yield and quality, and ability to capture the high diversity of RNAs in our samples, not to mention its competitive price. We will be switching over to using this product from now on!