What’s New at Quantabio?
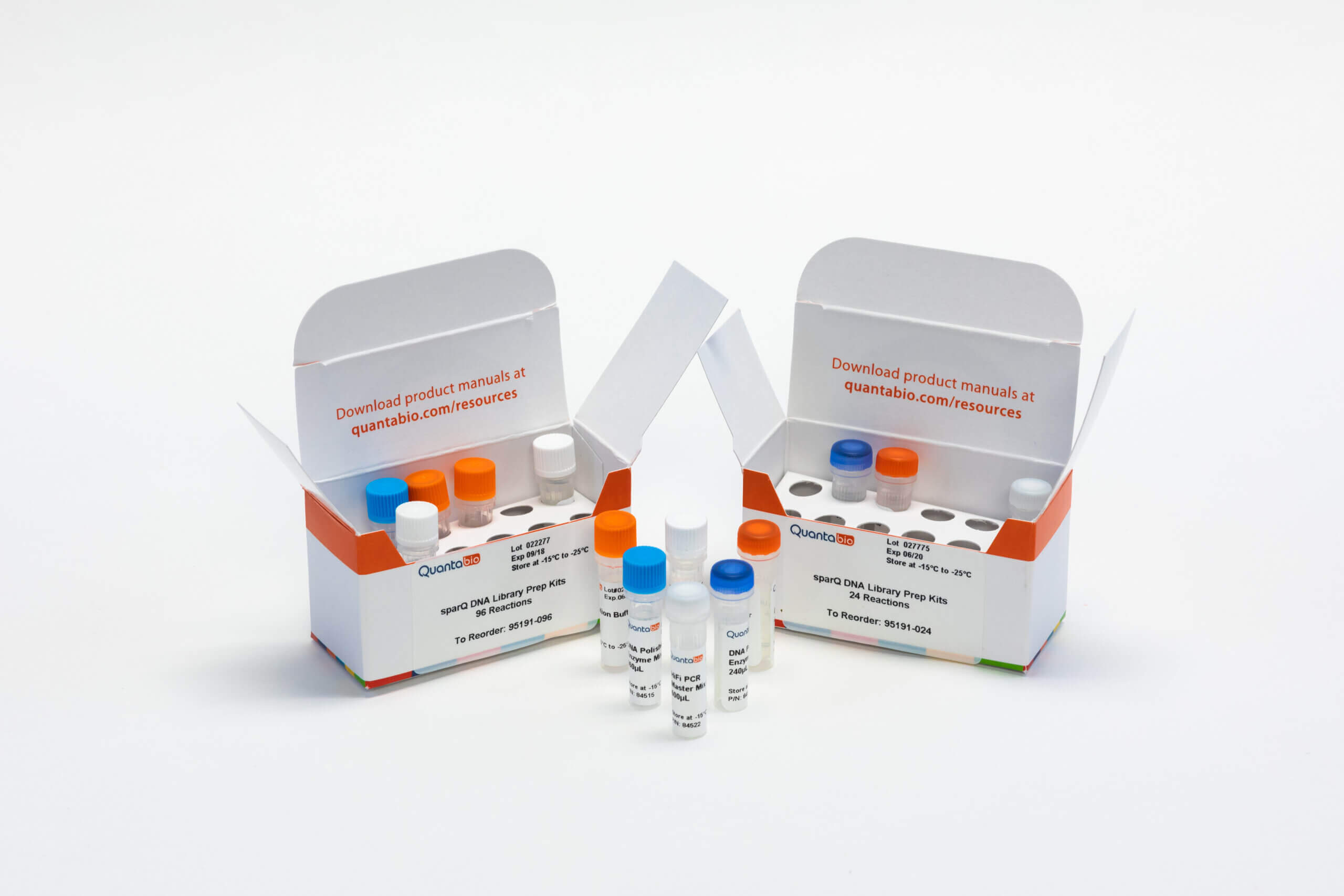
DNA Library Prep with sparQ
More Libraries. More Sequencing. More Savings. Quantabio is committed to reducing the library prep cost to help scientists unlock vital genetic information for a variety of sequencing applications.
repliQa HiFi ToughMix
Identifying the best PCR enzyme for library amplification in NGS for both short and long read sequencing, as published in Microbial Genomics
On-Demand Webinar
Watch our latest webinar “Crossing the limits of cDNA synthesis”

Avoid DNA polymerase, nucleic acid, and other common PCR inhibitors in crude blood samples with Quantabio ToughMixes.
Overcome the limitations of sensitivity and specificity in dried blood samples with Quantabio’s advanced ToughMixes.

Our ToughMix reagents withstand common PCR-suppressive compounds in plant & soil samples, such as phenols, polysaccharides, and humic acid.
& Reduce Costs
Quantabio offers complete library prep solutions for Illumina NGS platforms. High-quality reagents deliver unmatched efficiency and robust performance to ensure reliable and reproducible sequencing results while reducing total costs.


Size Selection

From product brochures and data sheets to trade publications and webinars, we have the most up-to-date information you’re looking for here.